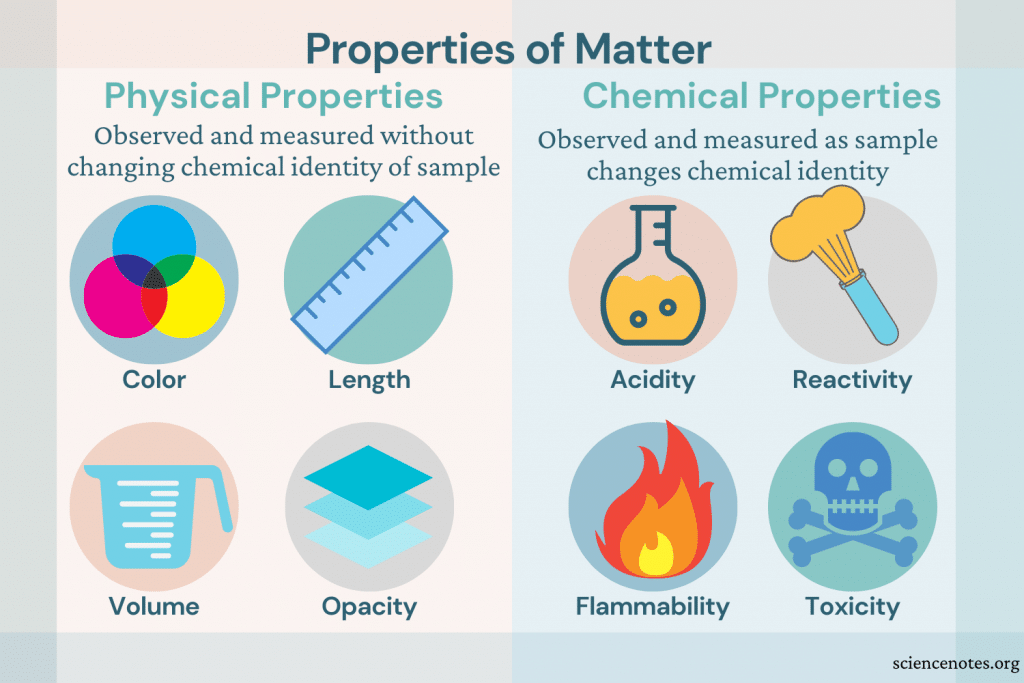
Whatever the reaction, it’s important to recognize that one or more new substances are formed! Burning wood is a common example of a chemical reaction because as the wood reacts with oxygen after it has been ignited, it creates smoke and ashes. The ways chemicals can react are many: two substances can combine to create a totally new one (Direct Combination), a substance can permanently break apart, separating into different substances (Decomposition), or maybe a substance combusts as it reacts to oxygen (Combustion) (see more types of reactions here). We bring in the term “reaction” when dealing with chemical changes because the chemicals involved actually react with one another to form a new substance (that’s why we call the parts in a chemical reaction the reactants). Signs of a physical change include:Ĭhemical changes, or reactions, involve chemicals getting rearranged in irreversible ways. Ripping paper is another example because while it may be in pieces, it’s still paper. 
A common example is of water freezing the liquid water becomes a solid, but it is still water. The molecular structure of the original substance(s) is the the same after the change, which means it is still the same substance. Physical changes involve chemicals getting rearranged in a new way, but without getting destroyed or irreversibly combined. Breathing, cooking, exercising, and more: we are surrounded by changes every day! But how do we keep straight which are physical and which are chemical?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
